LEARNING TARGET: STUDENTS CAN IDENTIFY AND PREDICT PRODUCTS FOR THE 5 TYPES OF EQUATIONS BY UNDERSTANDING HOW ATOMS INTERACT WITH EACH OTHER.
Identifying and Practicing Chemical Rxn
Below is an explanation of how to predict each of the 5 types of equations. Read each description carefully and take notes over the writing and the videos.
Synthesis or CombinationTo predict the products of a synthesis (combination) reaction you simply need to apply what you have learned in the bonding chapters. These equations will either produce an ionic compound, covalent compound, or an acid. You will need to evaluate the reactants to see what type of bond they will form.
Na + S --> Na2S The above reactants were a metal and non metal so we know it is ionic. Since the charge of sulfur is -2 then we need 2 sodium; using the criss cross method. |
DecompostionTo predict the products of a decomposition reaction you simply need to look at the product and know the components that make up that product.
MgO --> Mg + O2 Do NOT forget to make DIATOMIC MOLECULES for the "super 7". If any of the halogens or oxygen, nitrogen, or hydrogen break down into elemental form, you have to put a 2. |
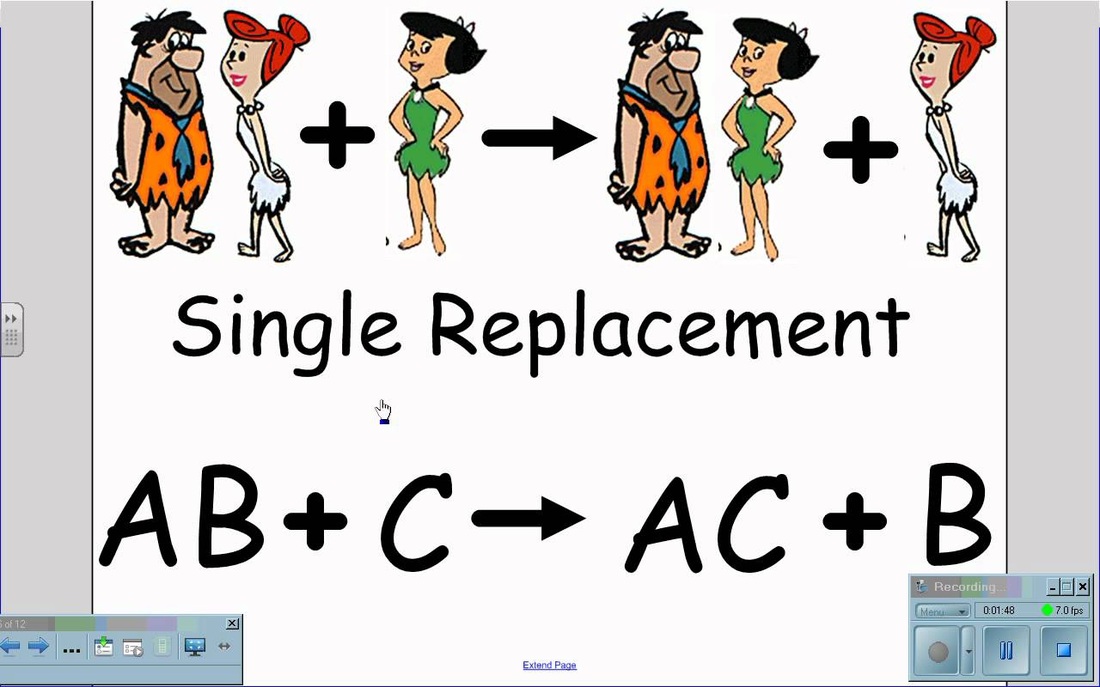
Single Replacement
To predict the products of a single replacement reaction simply SWITCH the ANIONS or the CATIONS (depending on which one is by itself). However they only switch if the element that is by itself in the reactants is MORE reactive than the one already bonded. You need to use the ACTIVITY SERIES OF THE ELEMENTS CHART below to tell you this information.
Li + BaCl2 ---> LiCl + Ba
This happens because Lithium is more reactive than barium (according to the chart below).
KF + Cl --> NO RXN
This has no reaction because fluorine is more reactive on the chart below than chlorine, so no reaction takes place. The reactants of a single replacement will always have 1 aqueous and either a sold or a gas for the single element.
Li + BaCl2 ---> LiCl + Ba
This happens because Lithium is more reactive than barium (according to the chart below).
KF + Cl --> NO RXN
This has no reaction because fluorine is more reactive on the chart below than chlorine, so no reaction takes place. The reactants of a single replacement will always have 1 aqueous and either a sold or a gas for the single element.
Oxidation and Single Replacements; evaluate the following equation.
Fe + Pb(NO3)2 --> Fe(NO3)3 + Pb
Notice that in the products we made Iron(III). This is because Iron can either be a +3 ion or a +2 ion (according to you Common Tables Chart) You can write EITHER +3 OR +2 and it would be right, if the elements is by itself in the reactants.
Fe + Pb(NO3)2 --> Fe(NO3)3 + Pb
Notice that in the products we made Iron(III). This is because Iron can either be a +3 ion or a +2 ion (according to you Common Tables Chart) You can write EITHER +3 OR +2 and it would be right, if the elements is by itself in the reactants.
Double Replacement
To predict the products of a double replacement reaction you simply switch the Cations and Anions in the reactants. This is always the case and does not have a chart to tell you if they switch like the single replacement. You just have to make sure that the products have CORRECT ionic formulas BEFORE you balance.
NaCl + MgO --> Na2O + MgCl2
Notice that in the products we made correct formulas, meaning sodium needed a 2 because of the -2 charge of oxygen and chlorine needed a 2 because of the +2 of magnesium. Double replacement reaction will always have 2 aqueous solutions for the reactants, 1 aqueous in the products and 1 that has a phase change.
NaCl + MgO --> Na2O + MgCl2
Notice that in the products we made correct formulas, meaning sodium needed a 2 because of the -2 charge of oxygen and chlorine needed a 2 because of the +2 of magnesium. Double replacement reaction will always have 2 aqueous solutions for the reactants, 1 aqueous in the products and 1 that has a phase change.
Combustion Reaction
To predict a combustion reaction you have to remember that there is always a carbon compound and oxygen in the reactants. In the products there is ALWAYS carbon dioxide and water produced.
This reaction is what happens in the kreb cycle in our body to give us energy and also the reaction that makes our cars run! Look at the two equations below for the examples.
This reaction is what happens in the kreb cycle in our body to give us energy and also the reaction that makes our cars run! Look at the two equations below for the examples.