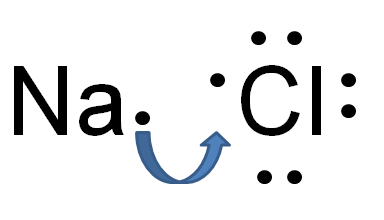LEARNING GOAL: STUDENTS CAN IDENTIFY, NAME, AND EXPLAIN PROPERTIES OF IONIC BONDS AND ACIDS AND BASES.
Ionic Bonding
|
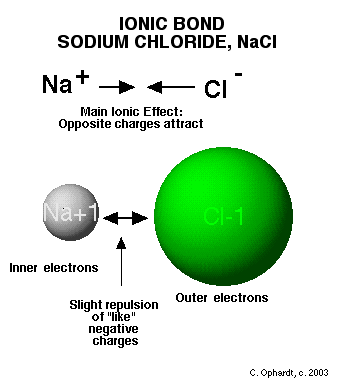
Ionic bonding is how ions are formed. When an element loses valence electrons or gains valence electrons they become either positive or negative ions (which we have already learned). After these elements are charged ions, they are attracted to each other, which is an ionic compound. We show how ions are made by using an electron dot structure. An electron dot structure is the elements symbol and then dots around the symbol that represent the VALENCE ELECTRONS. Electron dot structures are very useful in looking at bonding because valence electrons are the only ones important in bonding. Below is an example of an electron dot structure and the bond that forms the ions for sodium and chlorine which forms salt.
|
Important Vocabulary
|
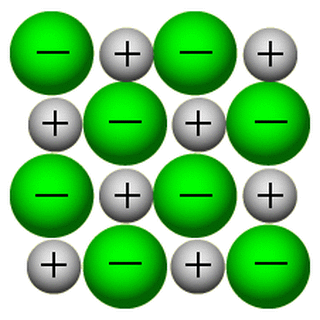
Ion Attraction and Network formation
WSQ and Worksheet Due Tuesday |