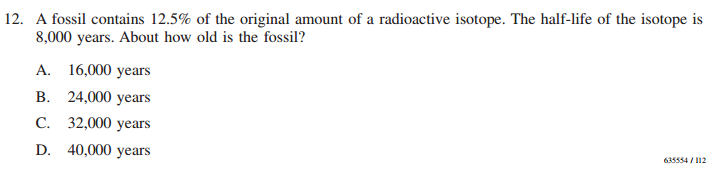Learning Target: Students can describe an isotope and calculate % abundance with practice and application.
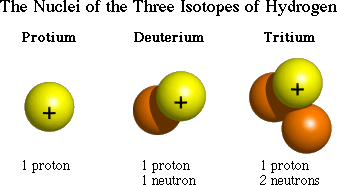
Isotopes are the SAME atoms that have a different number of NEUTRONS! ATOMIC MASS IS AN AVERAGE OF ALL ISOTOPES KNOWN ON EARTH! THAT IS WHY IS IT A DECIMAL.
Important Vocabulary
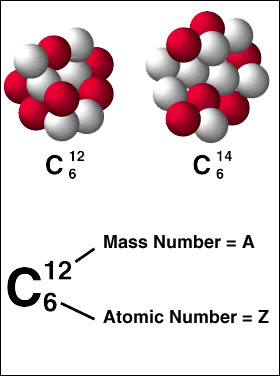
Writing in Isotope FormWhen you are writing isotopes, there is a special isotope form. When you are writing them with their symbols, you would do it as shown on the left carbon picture. When you are writing out the word, write out the name of the element and then the ATOMIC MASS. A common mistake is writing the number of neutrons; you need all the atomic mass (which is the number of protons and neutrons added together). See below for an example.
Carbon-12 (most abundance isotope of carbon) Carbon-14 (carbon with 2 EXTRA neutrons) |
% Abundance Calculation
So how do scientists get the ATOMIC MASS AVERAGE? Now that we know that the atomic mass of an element is a weighted average of the masses of its isotopes, you can now determine atomic mass based on the relative abundance of the isotopes. To do this you must know 3 values:
To calculate the atomic mass of an element, multiply the mass of each isotope by its natural abundance, expressed as a decimal, and then add the product. The resulting sum is the weighted average mass of the atoms of the element as they occur in nature.
Example Problem:
Element X has two natural isotopes. The isotope with a mass of 10.012 amu has a relative abundance of 19.91%. The isotope with a mass of 11.009 amu has a relative abundance of 80.09%. Calculate the atomic mass of this element.
- the # of stable isotopes of the element
- the mass of each isotope
- the natural percent abundance of each isotope
To calculate the atomic mass of an element, multiply the mass of each isotope by its natural abundance, expressed as a decimal, and then add the product. The resulting sum is the weighted average mass of the atoms of the element as they occur in nature.
Example Problem:
Element X has two natural isotopes. The isotope with a mass of 10.012 amu has a relative abundance of 19.91%. The isotope with a mass of 11.009 amu has a relative abundance of 80.09%. Calculate the atomic mass of this element.
|
Known
Isotope-10 mass = 10.012 amu relative abundance = 19.91% = 0.1991 Isotope-11 mass =11.009 amu relative abundance = 80.09% = 0.8009 |
Unknown
atomic mass of element X? |
Isotope-10 10.012 amu x 0.1993 = 1.993 amu
+
Isotope-11 11.009 amu x 0.8009 = 8.817 amu
10.8010 amu is the atomic mass of Element X
+
Isotope-11 11.009 amu x 0.8009 = 8.817 amu
10.8010 amu is the atomic mass of Element X
Radioactivity of Isotopes
|
|
|