Metallic Bonding
Learning Target: Students will be able to compare and contrast how electrons are shared in covalent and metallic bonding. Students will also be able identify important properties of metallic bonding.
We have already discussed Metallic Bonding and we know that it shares electrons. However this sharing is different from the sharing of covalent bonding because metallic bonding has a SEA OF ELECTRONS. Meaning that all electrons are shared with all other atoms around it, not a specific sharing like covalent. Two important properties of Metallic Bonding are that they make Alloys and have Interstitial Spaces.
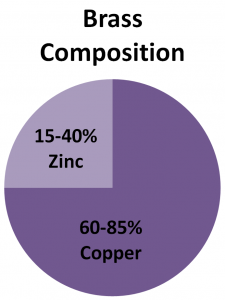
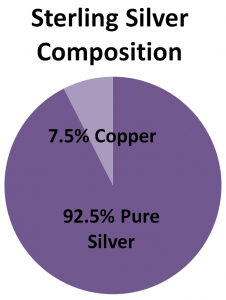
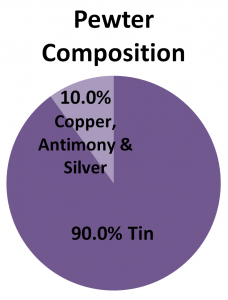
Alloys
Alloys are a combination of atoms on the Periodic Table that make a new substance. Below are three examples of alloys and their composition. Alloys are not on the Periodic Table.
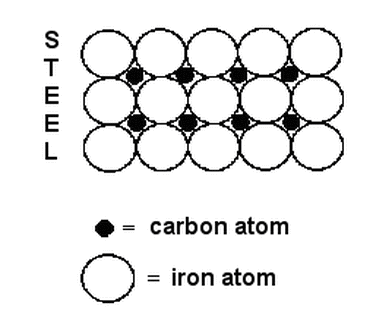
Interstitial Spaces
Interstitial space is the space between atoms in a metallic bond. A great example of interstitial space is the alloy of steel. Steel is one of the strongest substances because CARBON is in the interstitial spaces of the Iron atoms. This makes for a very dense solid that is very strong.