Monoatomic Ions and Oxidation Numbers
LEARNING TARGET: STUDENT CAN IDENTIFY MONATOMIC AND POLYATOMIC IONS.
|
Monoatomic ions are the elements that have either lost or gained electrons by bonding with another element. The Periodic Table helps us with all the monoatmoic ions in the "A groups" (the s and p-block), but it doesn't give us any information about the "B group" (the d-block). To be able to identify the charge of the B group we have to use the oxidation number. An oxidation number IS ALWAYS GIVEN IN THE NAME. We will discuss this more when we start naming ionic compounds. Below is a chart with the most common monoatomic ions, including some from the A group that we can find on the Periodic Table and others from the B group that we cannot. Notice that oxidation numbers are a Roman numeral. The Roman numeral tells the CHARGE of the monoatomic ion. So copper (I) has a positive one charge. ALL NUMBERS THAT ARE OXIDATION NUMBERS ARE POSITIVE. We will only see oxidation numbers in ionic bonding.
|
Polyatomic Ions
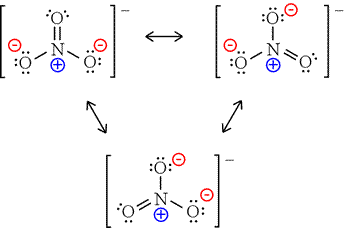
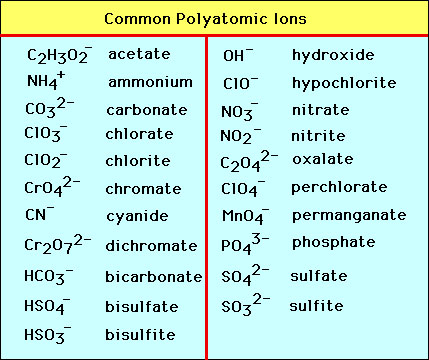
Polyatomic ions are a group of elements that are COVALENTLY BONDED, but are still not happy in terms of their electrons. Because of this, the molecule tries to give and take electrons just like an unhappy element. In the illustration below Nitrate is one nitrogen and three oxygen. However, the electrons have multiple places they can be located and therefore move giving the opportunity for other ions to bond with the molecule. An example of how nitrate would bond with sodium would be NaNO3. You must memorize the polyatomic ions! It becomes VERY important to know these when we start naming ionic and covalent bonds. Even though polyatomic ions are bonded covalently, we will only see polyatomic ions in ionic bonds.
Memorizing the Polyatomic Ions (Quiz Wednesday)
|
|
|
Quiz tomorrow over the following
- electron dot structure
- charges of monoatomic ions in A group
- formula writing for the elements
- polyatomic ion (name, charges, and formulas) use the chart below that was on the video