Learning Target: Students can identify the key regions, groups, and trends on the Periodic Table.
Periodic Table WSQ
Mendeleev's Periodic Law: "The physical and chemical properties of elements are periodic functions of their atomic masses."
Important Vocabulary
|
In class video |
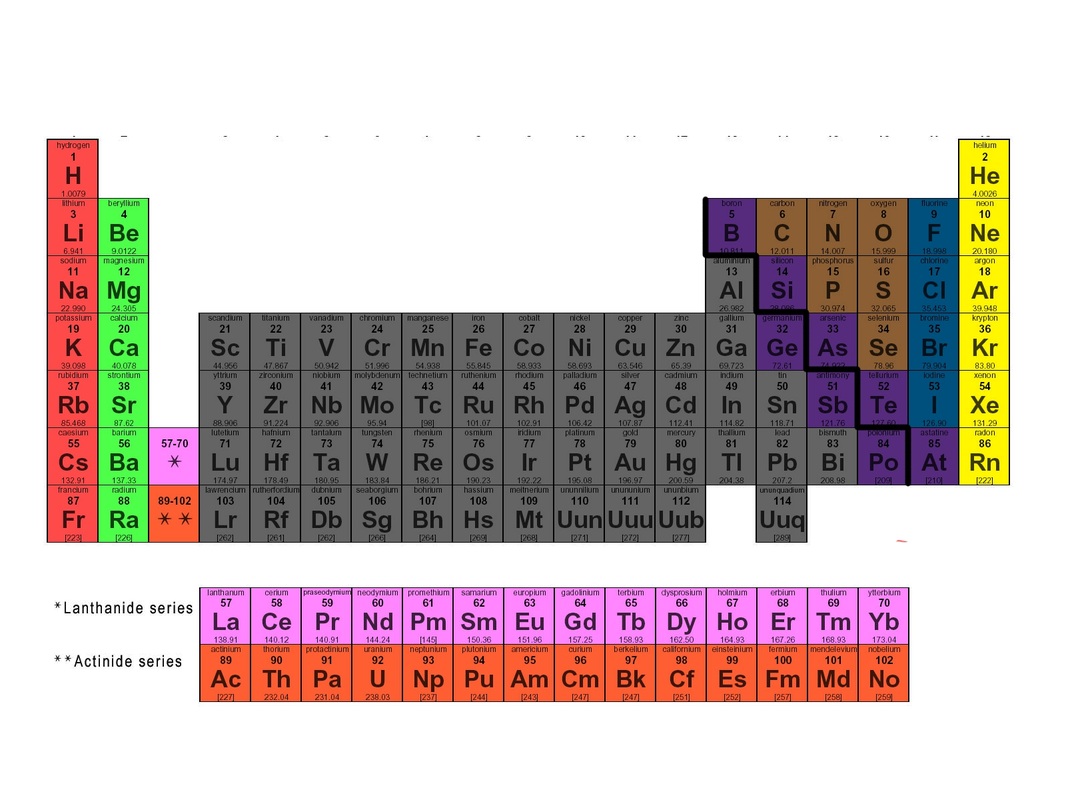
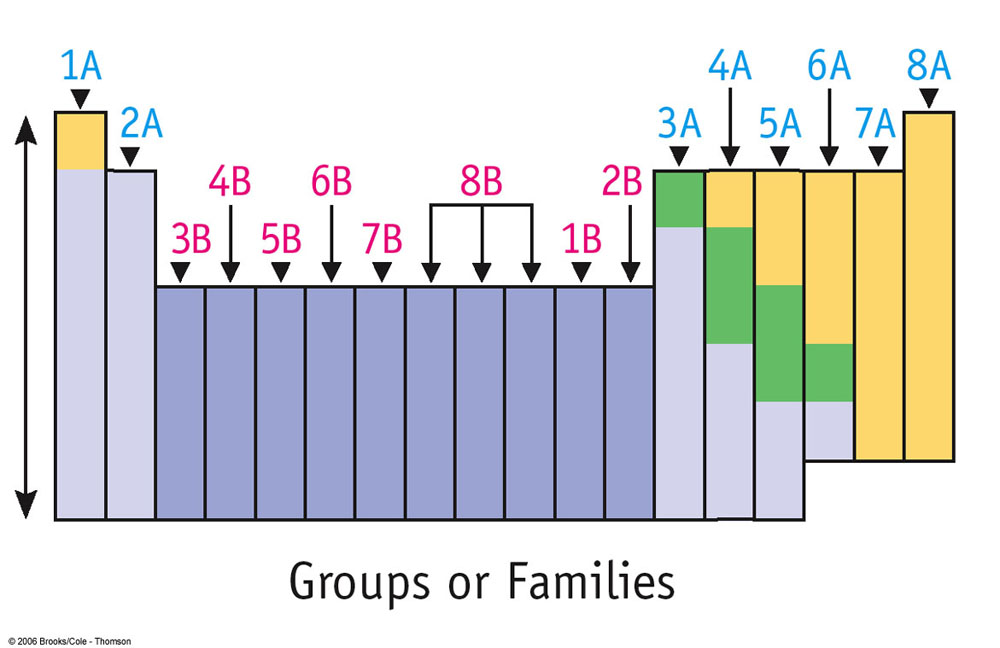
Regions of Periodic TableGroup 1A = Alkali Metals
Group 2A = Alkaline Earth Metals Group 3B-2B = Transition Metals Stair step in groups 3A-6A = Metalloids Group 7A = Halogens Group 8A = Noble Gases Elements 57-71 and 89-103 = Rare Earth BlocksGroups 1A and 2A = s-block
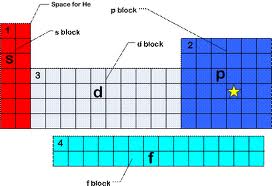
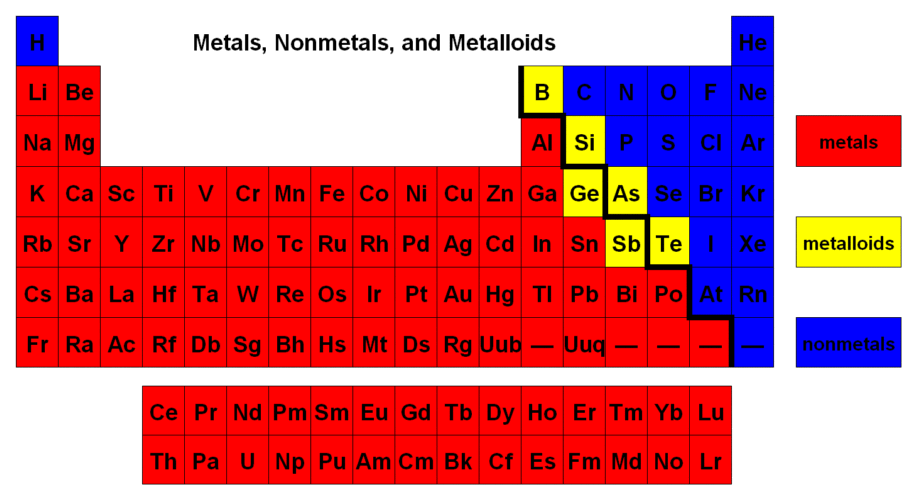
Groups 3B-2B = d-block Groups 3A-8A = p block Elements 57-71 and 89-103 = f-block Metals, non metals, and metalliods |
Rows and ColumnsColumns on the Periodic table are called "groups"
Rows on the Periodic table are called "periods" PeriodsThe periods on the periodic table are numbered 1-7 down the side. These indicate the number of shells the atom has.
Valence Electrons and IonsGroup A numbers are ALWAYS equal to the valence electrons.
Ions are formed when an atom gives or takes electrons from each other.
If you subtract group number from 8, you can get the CHARGE of the ion that the atom will form. |