Polarity/Dipole and Hybridization
LEARNING TARGETS:
-STUDENTS CAN EXPLAIN THE DIFFERENCE BETWEEN POLARITY AND DIPOLE.
-STUDENTS CAN IDENTIFY HOW HYBRIDIZATION GIVES SHAPE, POLARITY, AND A DIPOLE TO A MOLECULE.
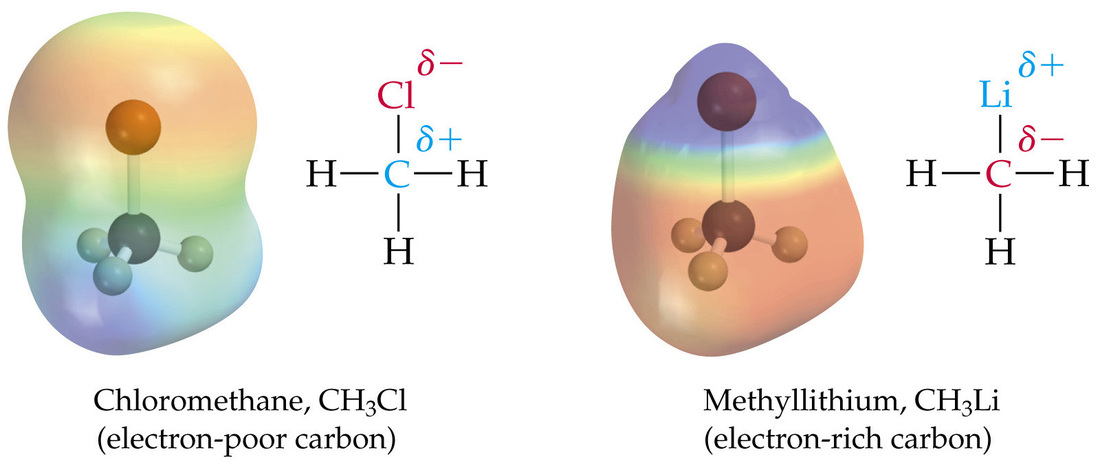
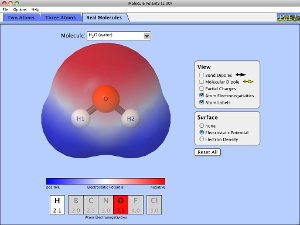
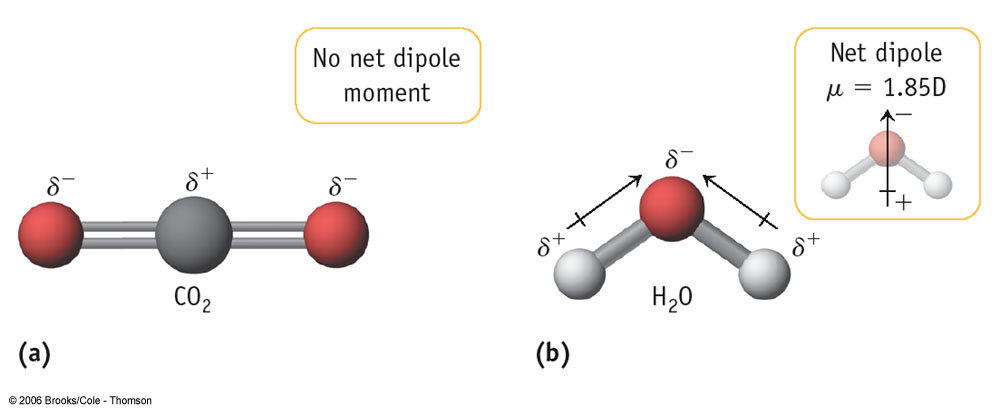
In covalent bonding you already know that electrons are being shared. However, these electrons are not always shared equally. When electrons between bonds are not shared equally we call this polarity. When the entire molecule has more electrons on one side than the other, we call this a dipole.
Polarity without a Dipole and with a Dipole
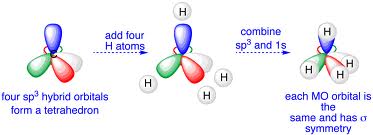
Hybridization
You will be excited you know that the information we learned about in the Electrons and Light unit is extremely useful for understanding hybridization. Hybridization mean a "hybrid" or combination of 2 orbitals (s,p,d,f) Below is an example of methane gas (CH4) and how it bonds using the p orbitals from carbon and the s orbitals from the 4 hydrogens. When these orbitals overlap, the electrons are free to go between to the atoms outer most shell. That is how a covalent bond "shares" electrons!