Learning Target: Students will be able to interpret a phase diagram in relation to pressure and temperature and understand the changing of matter.
Phase Changes flow chart Acitivity
Use the following words and organize them into a flow chart
- plasma
- gas
- liquid
- solid
- ionization
- vaporization
- melting
- deposition
- deionization
- sublimation
- freezing
- condensation
Gas Particles and Kinetic Theory
In the late nineteenth century, scientists developed the kinetic-molecular theory of matter to account for the behavior of the atoms and molecules that make up matter. The kinetic-molecular theory is based on the idea that particles of matter are always in motion. The theory can be used to explain the properties of solids, liquids, and gases in terms of the energy of particles and the forces that act between them.
PRESSURE, HEAT and Boiling Point
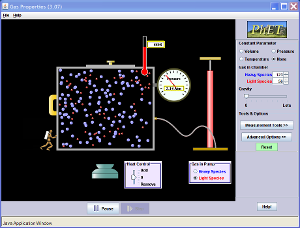
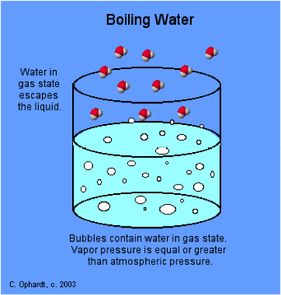
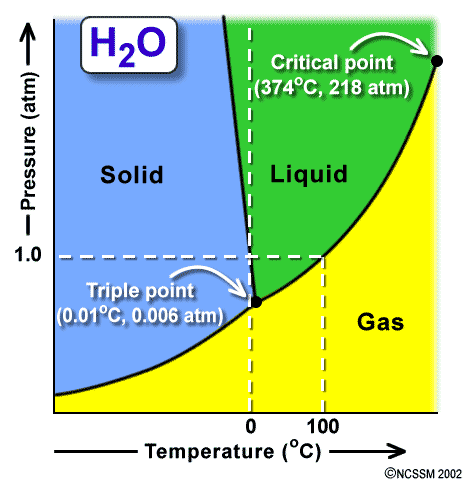
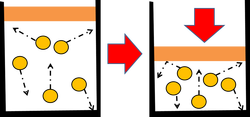
This is always a surprise to many of my students that we can boil water at room temperature by decreasing its pressure. To make water boil there are two factors that are important; pressure and heat. A phase diagram can tell us a lot about all state changes. One important feature of the phase diagram is that is tells us the different temperatures and pressures that a liquid can boil at. There is a phase diagram for every type of substance.
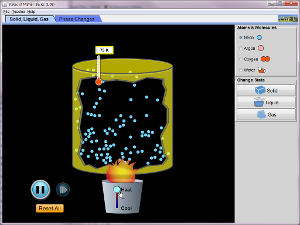
Liquids have a definite volume as shown above, but no definite shape. This is because of the flowing molecules or in Bromine (Br) and Mercury (Hg) case, flowing atoms. Liquid molecules (or atoms) are spaced further apart than solid, but close together than a gas. If you want to change a liquid into a gas you have to speed up the molecules (or atoms) to get them to spread out. If you want to change a liquid into a solid, you have to slow down the molecules (or atoms). These changes can be graphically shown on a Phase Diagram. We are going to learn how to interpret these graphs and see the information about STATE CHANGE on them. Watch the video below to learn about the graphs properties.
- Pressure
- Boiling Point
- Surface Tension
- Hydrogen bond
- Fluid
Important Vocabulary
- Triple point
- Phase Diagram
- Phase Change
- Super-critical Fluid
- Absolute Zero
- Kinetic Theory
- Sublimation
- Deposition
- Condensation
- Vaporation (evaporation)
Phase Diagram Questions
- The evaporation of liquid water from the surface of Earth is an important step in the water cycle. How do water molecules obtain enough kinetic energy to escape into the gas state?
- What is the difference between evaporation and vaporation?
- How does the kinetic-molecular theory explain the pressure exerted by gases?
- What happens to gas particles when a gas is compressed?
- What happens to gas particles when a gas is heated?
- Molecules of hydrogen escape from Earth, but molecules of oxygen and nitrogen are held to the surface and remain in the atmosphere. Explain.