EVERYTHING YOU LEARNED ABOUT DRAWING ATOMS IN PHYSICAL SCIENCE WAS A LIE!!!
LEARNING TARGET: STUDENT WILL BE ABLE TO UNDERSTAND HOW ELECTRONS ORBIT AROUND THE NUCLEUS OF AN ATOM ON AN X AND Y AXIS AND SHOW THE FILLING OF ELECTRONS IN AN AUFBAU DIAGRAM.
THERE ARE 3 WAYS TO SHOW HOW ELECTRONS FILL THE SHELLS OF AN ELECTRON.
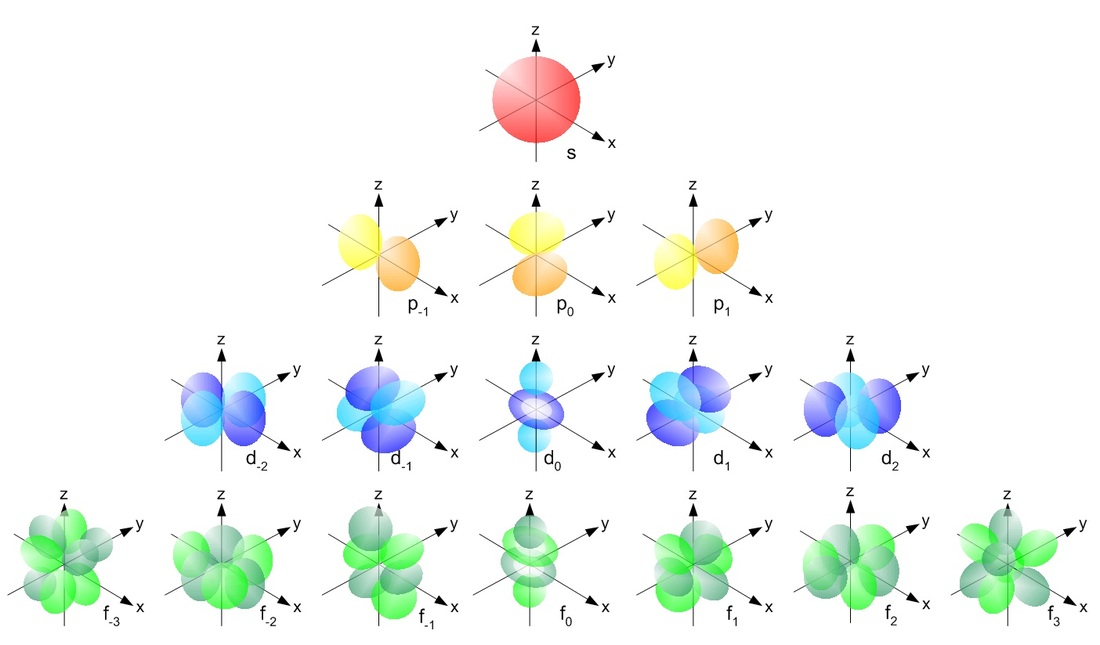
- DRAW THEM ON AN X-Y-Z AXIS WITH ORBITAL SHAPES
- WRITE THEM IN SENTENCE FORM WITH ELECTRON CONFIGURATION
- DRAW ARROWS IN AN AUFAU DIAGRAM TO SHOW ELECTRONS IN CHART FORM
Atomic Orbitals
|
|
|
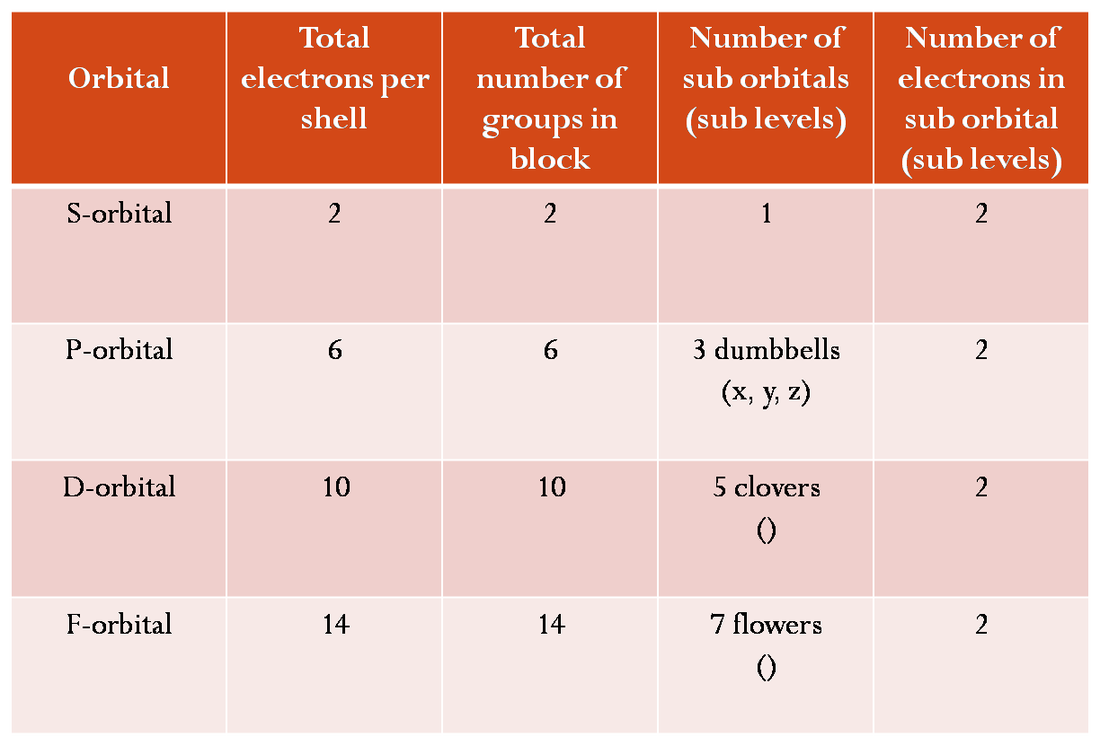
You have probably gathered that the model of the atom that you learned in Physical Science is not true. You are correct. The atoms DO NOT orbit around the nucleus in nice circular patterns that increase in size. They are actually made of four main shapes; see chart below
Electron Configuration
Electron configuration shows in "sentence form" how electrons are filling the shell of an atom.
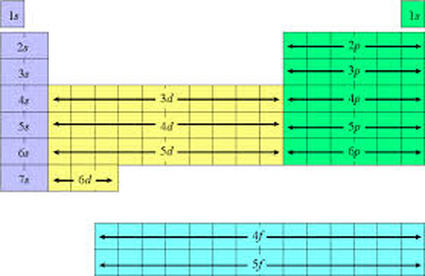
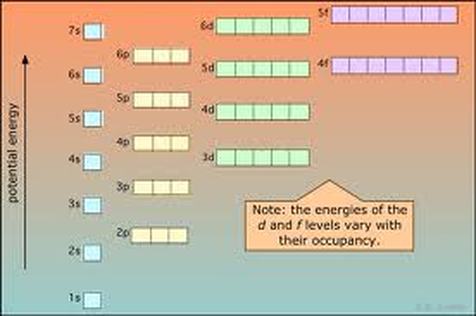
Aufbau Diagram
Aufbau Diagrams show the filling of the different orbitals (learned above) in chart form. The drawing of orbitals and the filling of the aufbau diagram are exactly the same.